The open ocean system in the instrumental period
- This page is part of the topic Marine biology in the instrumental period
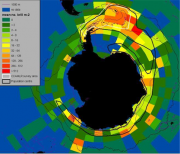
The area covered by winter sea ice in the Southern Ocean has not changed significantly over the past decades suggesting that the impact of global warming on Antarctic ecosystems is not as severe as it is in the Arctic. There the sea ice cover is declining in both thickness and extent at a rapid rate, profoundly affecting the structure and functioning of Arctic marine ecosystems, particularly mammal and bird populations. For a comprehensive and most recent review with additional relevant literature see Nichol (2008). In the Antarctic, comparable shrinking of the winter ice cover has occurred only along the western side of the Peninsula and adjoining seas. This is a relatively small region but home to the well-known whale-krill-diatom food chain. Based on data shown in Figure 4.46, Atkinson et al. (2008[1]) calculated that 70% of the total krill stock resides in the sector 0°-90°W, a region characterised by rapid regional changes both in water temperature (Meredith and King, 2005[2]; Whitehouse et al., 2008[3]) and winter sea ice cover (Parkinson, 2004[4]).
Following near-extinction of the whale populations, the krill stock was expected to increase as a result of release from grazing pressure. Although predation pressure by seals and birds increased, the total biomass remained only a few percent of that of the former whale population. About 300,000 blue whales alone were killed within the span of a few decades equivalent to more than 30 million tonnes of biomass. Most of these whales were killed on their feeding grounds in the southwest Atlantic in an area of at most 2 million km2 (10 % of the entire winter sea ice cover), which translates to a density of one blue whale per 6 km2. Today’s whale watchers would be thrilled. A 100 tonne blue whale (adults weigh 150 tonnes) contains about 10 tonnes of carbon, so the biomass of the whales on their feeding grounds would have amounted to 1.5 g C m-2 which is equivalent to the average coastal zooplankton biomass. Adding the biomass of krill estimated to have been annually eaten by the whales (150 million tonnes) to the m2 calculation, we get 12 g C m-2 just for blue whales and their annual food intake. This number is equivalent to the biomass of an average phytoplankton bloom or, to take an example of more familiar grazers, to 240 cows of 500 kg each grazing on one km2 of meadow.
The actual krill stock, from which the 150 million tonnes were being eaten, will have been at least three times higher prior to whaling. The magnitude of primary production required them to fulfil their food demands at the trophic transfer rule of thumb (10:1) would be around 300 g C m-2 yr-1 which is about that estimated for the North Sea, hence this does not leave much scope for other grazers such as protozoa and copepods. What percentage of the production was exported then from the surface through the mesopelagial habitat and ultimately to the deep-sea benthos, hence also sequestered as carbon, is an interesting but open question.
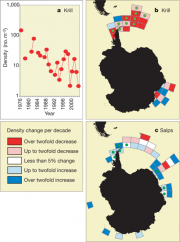
The above calculations indicate that krill stocks in the whale feeding grounds were close to the carrying capacity of the ecosystem prior to whaling. That being the case, when less krill was eaten by whales, enabling more krill to survive, many of the survivors would have starved. This would explain why a krill surplus, at least equivalent to the amount annually eaten by whales, was not recorded. At their former high stock sizes, and given the tendency of krill schools to appear at the very surface and discolour the water, they were commonly observed from ship decks, as noted for example by the scientists of the Discovery cruises (Hardy, 1967[6]). Since that time, despite a significant increase in the numbers of observers, from cruise ships to research vessels, krill swarms are now rarely seen from ships decks. A thorough statistical assessment of all net catches has been carried out for different sectors of the Southern Ocean. The analysis suggests a 38 - 81% decline in krill stocks of the southwest Atlantic accompanied by an increase in salp populations (Figure 4.47, Atkinson et al., 2004[5]; Ross et al., 2008[7]). The extent of the krill decline and the underlying factors are under vigorous debate (Ainley et al., 2007[8]; Nicol et al., 2007[9]), because of difficulties in unravelling the effects of industrial whaling from those of sea ice retreat; there are also discrepancies between the abundances of krill as measured by net and acoustic methods, and enormous intra-annual as well as spatial variability has to be considered (Hewitt et al., 2003[10]; Saunders et al., 2007[11]). However, a significant negative correlation between krill density (30°W to 70°W) and mean sea surface temperature at South Georgia has been found for the period 1928-2003, which implies a large-scale response not only of krill but of the entire open ocean ecosystem to climate change (Whitehouse et al., 2008[3]).
If former krill stocks were close to the carrying capacity provided by primary production, then a decrease in grazing pressure should have resulted in a “phytoplankton surplus”, but there is also little evidence for that. Unfortunately, a comparison with phytoplankton stocks recorded during the Discovery era is not possible because the methods used at that time soon became obsolete. Nevertheless, the impression gained by the Discovery scientists is one of large diatom stocks: “...extremely rich production, which will probably be found to exceed that of any other large area in the world …” (Hart, 1934[12]). The Discovery scientists were familiar with North Sea phytoplankton, which today has much higher biomass levels than those recorded for the Scotia Sea in recent decades. That comparison makes it likely that phytoplankton production has indeed decreased with that of krill stocks, a conclusion supported by the increase in the salp population. In contrast to krill, which are equipped to deal with the characteristically spiny and heavily silicified diatoms of the Southern Ocean, salps are adapted to feed on the lower biomass concentrations typical of the iron-limited microbial food webs. Their encroachment into the former krill habitat is an indication of declining phytoplankton, in particular diatom stocks.
A decline in phytoplankton concentrations can be explained by a corresponding decline in the supply of iron. There is reason to believe that the reduction in sea ice formation has resulted in a decrease in iron input from the continental margins of the Western Peninsula. In contrast, the simultaneous retreat of glaciers should have increased run-off, and possibly also iron input, from the land along the coasts of the Peninsula and adjacent islands. Comparisons of the chlorophyll concentrations recorded by the CZCS satellite of the 1980s with those from the current SeaWifs satellite indicate a decline along the Antarctic ice edge and particularly in the Scotia Sea, the only region of the globe where production has declined, but no major change along the coast (Gregg and Conkright, 2002[13]). Production was found to have increased by 50%, off the Patagonian shelf, so it is also possible that the wind field transporting Patagonian dust from mud fields laid bare by retreating glaciers has changed, reducing the aeolian iron supply to the Scotia Sea. Also, the westerlies have intensified and would carry more dust. Whatever the mechanism, a reduction in phytoplankton biomass can only be explained by a corresponding reduction in iron supply combined with light limitation by deep mixed layers and heavy grazing pressure on phytoplankton stocks. Recently, five mesoscale, in situ iron fertilization experiments, carried out in the Pacific and southeast Atlantic Sectors of the Southern Ocean, have unambiguously demonstrated that plankton biomass is limited by iron availability. It follows that the higher productivity of coastal regions, including the southwest Atlantic, is maintained by input of iron supplied from land-masses, and from the sediments by deep mixing and upwelling along the continental margin. The presence of excess nutrients in these regions allows the assumption that iron supply limits productivity over most of the year throughout the Southern Ocean. The ramifications of this finding for the structure and functioning of Antarctic ecosystems have yet to be adequately explored, particularly because ongoing global change will affect coastal hydrography and hence the supply of iron.
An alternative but not mutually exclusive explanation for the phytoplankton decline can be a decrease in the rate of recycling of the iron entering the system. It is now well established that primary production in microbial food webs is based on recycling by grazers feeding on pico- and nano-phytoplankton, which are in turn eaten by predators such as ciliates and copepod larvae. The latter are the preferred prey of copepods, whereas filter-feeding salps consume all the components en masse. In the Southern Ocean the microbial community is characteristic of the iron-limited HNLC (high nutrient, low chlorophyll) area, where chlorophyll concentrations remain below 0.5 mg-Chl/m2 throughout the year. These regions support a surprisingly high zooplankton biomass, comprising slow growing copepods and fast-growing salps, throughout the year, suggesting that they are an integral part of a recycling system that also regenerates iron in addition to ammonium (Barbeau et al., 1996[14]).
Local increases in production above this level are invariably due to accumulation of diatoms and Phaeocystis colonies, and will be caused by input of new iron, whether from above or from below. The fate of these diatom blooms is under debate: are they consumed and their nutrients recycled in the surface or sub-surface layer, or does a significant portion sink to greater depths or to the sea floor? The latter fate is of particular interest in the light of proposals for large-scale ocean fertilization to sequester atmospheric CO2.
Given the high densities of the former krill stocks, their rate of recycling will have been as effective as that in the microbial food web today, but on a much larger scale. Predation by whales will have contributed significantly to the iron recycling pool. That is because, except in the case of pregnant and lactating females, whales convert krill protein into blubber (hydrocarbons), so retaining energy but excreting nutrients, including iron. Whale faeces are liquid and rise to the surface where they are likely to have released iron, thereby increasing the efficiency of recycling. Krill also have a high (50%) lipid content, and krill excretion releases large amounts of iron (Tovar-Sanchez et al., 2007[15]). It follows that the exceptionally productive ecosystem characterised by the food chain of the giants was maintained by the recycling of iron by krill and whale feeding. An alternative, but mutually inclusive hypothesis in which large whale stocks promoted the development of large stocks of their prey (krill) by dispersing them over a larger area has been recently suggested.
It is now acknowledged that large terrestrial herbivores (megafauna) condition ecosystems by promoting a vegetation cover conducive to their demands, e.g. grassland instead of forest by elephants. The removal of those herbivores leads to profound changes in landscape. Similarly, the top predators in lakes can determine the structure of the ecosystem down to the composition and biomass of the phytoplankton. Predation pressure on upper trophic levels is propagated down the food web by mechanisms known as trophic cascades. Although the effects of top-down control have been demonstrated for shallow benthic environments from many coastal regions, comparable mechanisms are only now coming to light from planktonic ecosystems, such as the reported worldwide increase in gelatinous plankton after removal of dominant fish stocks. Whether such changes can cascade down to the level of phytoplankton is not known. We simply do not know what effect the removal of whales (and seals) through hunting had on the ocean ecosystem around Antarctica. The belief that marine phytoplankton productivity is determined primarily by bottom-up driving forces is entrenched in the marine biological literature, but why the interaction between marine plankton and whales around Antarctica should be fundamentally different from that between their non-polar lake counterparts has yet to be addressed. The fact is that the processes driving annual cycles of phytoplankton production, biomass and species composition in the marine environment remain largely unknown. It is time to explore new approaches.
The linkages between phytoplankton and bacterioplankton in the Southern Ocean are not well understood. The timing of spring phytoplankton blooms and bacterioplankton activities are not necessarily linked, as they are in other pelagic systems, though there may just be a lag in this linkage that is longer than in lower latitude systems (Ducklow et al., 2007[16]). Even less is known of the relationships between phytoplankton species and bacterioplankton species composition (e.g. whether particular bacteria are associated with diatom vs. cryptophyte phytoplankton). Considerations of phytoplankton primary productivity, linkages to CO2 drawdown, and grazer populations are also linked to phytoplankton species composition. Phytoplankton species are susceptible to changes in sea ice duration and position of the ice edge, and shifts in water column properties such as the depth of the mixed layer. Shifts in phytoplankton species composition from diatom-dominated communities to more diverse communities dominated by cryptomonads and flagellates occurs following the ice edge retreat and spring diatom bloom in the Western Antarctic Peninsula (Moline and Prezelin, 1996[17]), and will be (or already are) potentially more common occurrences as a result of warming in this region (Clarke et al., 2007[18]). The linkages between phytoplankton and zooplankton populations are tight, as krill tend to dominate the zooplankton assemblages when diatoms are abundant, and zooplankton dominance can shift to salp dominance when the community is cryptomonad or flagellate dominated. One study in the Western Antarctic Peninsula recently reported that there has been a shift from krill-dominated waters to salp dominance since 1999 This trend may also be potentially representative of the longer term trends referred to above.
Acquiring a mechanistic understanding of the structure and functioning of the ecosystems surrounding Antarctica is a prerequisite for predicting their performance under the influence of global warming. Hypothetical conceptual frameworks of relevant mechanisms need to be developed that can be tested by comparing intact ecosystems with those where top-predators have been depleted both regionally and, where baseline data are available, temporally. Satellite data have vastly extended the scales accessible to such regional studies. Larger scale in situ iron fertilization experiments open up an exciting new avenue to study the effects of bottom-up versus top-down factors on higher trophic levels, and if carried out over several years, also on krill populations and on the underlying deep sea and benthos. Such experiments provide an ideal background to study the relationship between ecology and biogeochemistry at the species level, which in turn will improve interpretation of sedimentary proxies, in particular microfossils, for reconstruction of past climate change. Conceptual frameworks emerging from field studies and experiments can be explored, tested and refined with new generations of 4D mathematical models.
Iron fertilization experiments
It has been suggested that one way in which the rise of carbon dioxide in the atmosphere may be mitigated is to fertilise the ocean with iron so as to stimulate the production of plankton and hence the draw-down of carbon dioxide from the atmosphere into the ocean (Boyd et al., 2007). These ideas are based on the results of a limited number of experiments in which different parts of the ocean, including the Southern Ocean, were seeded with iron (Boyd et al., 2007b[19]). This current debate (e.g. see Oceanus, 24 June 2009, http://www.whoi.edu/oceanus/viewArticle.do?id=34167) may at some time shift its focus to exploring how to maximise the efficiency of the process and minimise harmful side effects. The hypothesis could be tested by a new generation of iron fertilization experiments carried out at larger scales and longer periods on the former whale feeding grounds. Given the apparent high rates of krill decline and a steady southward encroaching ocean warming there is a pressing need to develop an integrated understanding of how this ecosystem functioned not only in the recent past but also in the glacial ocean, in order to predict future changes in the pelagic and underlying benthic ecosystems around Antarctica. In situ iron fertilization experiments provide one methodology for testing ecosystem models, by enabling the study of interactions within ecosystems with a full complement of grazers and pathogens. The effect of iron fertilisation on higher trophic levels will depend on the locality and duration of the experiment. A regional survey of the underlying benthos prior to fertilization would yield a baseline to monitor possible changes in this ecosystem. Preliminary surveys of the deep-sea benthos of the Peninsula region have shown the presence of communities with high biomass and species numbers (Brandt et al., 2007[20]) but their areal extent is not known. Deep carbon export flux has been shown to be above global average and to have a high regional variability in the Southern Ocean (Boyd and Trull, 2007[21]; Sachs, 2008[22]). Since fertilization will be carried out offshore, it is quite unlikely that shelf and coastal benthos are significantly affected.
An added incentive to carrying out such experiments is that they would offer an ideal training ground for the kind of large-scale international, interdisciplinary research taking a whole Earth System Science approach to investigating global change. Such large-scale experiments would not only provide a wealth of new insights into the structure and functioning of pelagic and underlying benthic ecosystems. They could also provide more reliable data for parameterising current and new coupled ecological-biogeochemical ocean-circulation models for use in assessing the Southern Ocean as a sink for anthropogenic CO2. There is a concern that the incentive offered by the carbon credit market could result in excessive fertilization which could lead to unacceptable harm to Southern Ocean ecosystems (Chisholm et al., 2001[23]). Three United Nation bodies, the Intergovernmental Oceanographic Commission (IOC), the International Maritime Organization (IMO), and the Convention on Biological Diversity (CBD) agreed that proposals to use ocean fertilization to sequester carbon in the ocean give cause for concern due to unknown negative impact to the ecosystems (http://ioc3.unesco.org/oanet/OAdocs/INF1247-1.pdf). The two latter organizations recently argued that large operations are currently not justified and should not be allowed (http://www.cbd.int/decisions/?m=COP-09&id=11659&lg=0, http://www.maritime-connector.com/NewsDetails/2203/lang/.wshtml, http://www.ioccp.org). Scientists emphasized the necessity for independent research on small scale fertilisation studies (Buesseler et al., 2008[24]). In addition it must be considered that once done such large global experiments with unknown outcomes would be very difficult or impossible to reverse.
References
- ↑ 1.0 1.1 Atkinson, A., Siegel, V., Pakhomov, E.A., Rothery, P., Loeb, V., Ross, R.M., Quetin, L.B., Schmidt, K., Fretwell, P., Murphy, E.J., Tarling, G.A. and Fleming, A.H. 2008. Oceanic circumpolar habitats of Antarctic krill, Mar. Ecol. Prog. Ser, 362, 1-23.
- ↑ Meredith, M.P. and King, J.C. 2005. Rapid climate change in the ocean west of the Antarctic Peninsula during the second half of the 20th century, Geophys. Res. Lett., 32, L19604. (doi: 10.1029/2005GL024042)
- ↑ 3.0 3.1 Whitehouse, M.J., Meredith, M.P., Rothery, P., Atkinson, A., Ward, P. and Korb, R.E. 2008. Rapid warming of the ocean at South Georgia, Southern Ocean during the 20th Century: forcings, characteristics and implications for lower trophic levels, Deep-Sea Research I, 55, 1218-1228.
- ↑ Parkinson, C.L. 2004. Southern Ocean sea ice and its wider linkages: insights revealed from models and observations, Antarctic Science, 16, 387-400.
- ↑ 5.0 5.1 5.2 Atkinson, A., Pakhomov, E., Rothery, P., Siegel, V. 2004. Long-term decline in krill stocks and increase in salps within the Southern Ocean, Nature, 432 (7013), 100-103.
- ↑ Hardy, A. 1967. Great waters. Harper and Row, New York, 542 pp.
- ↑ Ross, R.M., Quetin, L.B., Martinson, D.G., Iannuzzi, R.A., Stammerjohn, S.E. and Smith, R.C. 2008. Palmer LTER: Patterns of distribution of five dominant zooplankton species in the epipelagic zone west of the Antarctic Peninsula, 1993-2004, Deep-Sea Res. II, 55, 2086-2105.
- ↑ Ainley, D.G., Ballard, G., Ackley, S., Blight, L., Eastman, J.T., Emslie, S.D., Lescroel, A., Olmastroni, S., Townsend, S.E., Tynan, C.T., Wilson, P. and Woehler, E. 2007. Paradigm lost, or is top-down forcing no longer significant in the Antarctic marine ecosystem? Antarctic Sci., 19, 283-290.
- ↑ Nicol, S., Croxall, J., Trathan, P., Gales, N. and Murphy, E. 2007. Paradigm misplaced ? Antarctic marine ecosystems are affected by climate change as well as biological processes and harvesting, Antarctic Sci., 19, 291-295 .
- ↑ Hewitt, R.P., Demer, D.A. and Emery, J.H. 2003. An 8-year cycle in krill biomass density inferred from acoustic surveys conducted in the vicinity of the South Shetland Islands during the austral summers of 1991-1992 through 2001-2002, Aquatic Living Resources, 16, 205-213.
- ↑ Saunders, R.A., Brierley, A.S., Watkins, J.L., Reis, K., Murphy, E.J., Enderlein, P. and Bone, D.G. 2007. Intra-Annual variability in the density of Antarctic krill (Euphausia superba) at South Georgia, 2002-2005: within-year variation provides a new framework for interpreting previous 'annual' estimates of krill densitiy, CCAMLR Science, 14, 27-41.
- ↑ Hart, T.J. 1934. On the phytoplankton of the South-West Atlantic and the Bellinghausen Sea, 1929-31, Discovery Reports, 8, 1-268.
- ↑ Gregg, W.W. and Conkright, M.E. 2002. Decadal changes in global ocean chlorophyll, Geophys. Res. Lett., 29, 21-24.
- ↑ Barbeau, K., Moffett, J.W., Caron, D.A., Croot, P.L. and Erdner, D.L. 1996. Role of protozoan grazing in relieving iron limitation of phytoplankton, Nature, 380, 61-64.
- ↑ Tovar-Sanchez, A., Duarte, C.M., Hernandez-Leon, S. and Sanudo-Wilhelmy, S.A. 2007. Krill as a central node for iron cycling in the Southern Ocean, Geophys. Res. Lett., 34, (L11601, doi:10.1029/2006GL029096).
- ↑ Ducklow, H.W., Baker, K., Martinson, D.G., Quentin, L.B., Ross, R.M., Smith, R.C., Stammerjohn, S.E., Vernet, M. and Fraser, W. 2007. Marine pelagic ecosystems: the West Antarctic Peninsula, Phil. Trans. R. Soc. B, 362, 67-94.
- ↑ Moline, M.A. and Prezelin, B.B. 1996. Palmer LTER 1991-1994: Long-term monitoring and analyses of physical factors regulating variability in coastal Antarctic phytoplankton biomass, in situ productivity and taxonomic composition over subseasonal, seasonal and interannual time scales phytoplankton dynamics, Mar. Ecol. Prog. Ser., 145, 143-160.
- ↑ Clarke, A., Murphy, E.J., Meredith, M.P., King, J.C., Peck, L.S., Barnes, D.K.A. and Smith, R.C. 2007. Climate change and the marine ecosystem of the western Antarctic Peninsula, Phil. Trans. R. Soc. B, 362, 149-166.
- ↑ Boyd, P.W., Jickells, T., Law, C.S., Blain, S., Boyle, E.A., Buesseler, K.O., Coale, K.H., Cullen, J.J., De Baar, H.J.W., Follows, M., Harvey, M., Lancelot, C., Levasseur, M., Owens, N.P.J., Pollard, R., Rivkin, R.B., Sarmiento, J., Schoemann, V., Smetacek, V., Takeda, S., Tsuda, A., Turner, S. and Watson, A.J. 2007b. Mesoscale iron enrichment experiments 1993-2005: Synthesis and future directions, Science, 315, 612-617.
- ↑ Brandt, A., Gooday, A.J., ET AL. 2007. First insights into the biodiversity and biogeography of the Southern Ocean deep sea, Nature, 447(7142), 307-311.
- ↑ Boyd, P.W. and Trull, T.W. 2007. Understanding the export of biogenic particles in oceanic waters: Is there consensus? Progress in Oceanography, 72, 276-312.
- ↑ Sachs, O. 2008. Benthic organic carbon fluxes in the Southern Ocean: regional differences and links to surface primary production and carbon export, Ber. Polarforsch. Meeresforsch., 578, 1-143.
- ↑ Chisholm, S., Falkowski, P. and Cullen, J. 2001. Dis-crediting ocean fertilization, Science, 294, 309-310
- ↑ Buesseler, K.O., Doney, S.C., Karl, D.M., Boyd, P.W., Caldeira, K., Chai, F., Coale, K.H., De Baar, H.J.W., Falkowski, P.G., Johnson, K.S., Lampitt, R.S., Michaels, A.F., Naqvi, S.W.A., Smetacek, V., Takeda, S. and Watson, A.J. 2008. Ocean Iron Fertilization--Moving Forward in a Sea of Uncertainty, Science, 319, 162.